In recent years there have been important developments regarding the availability of highly innovative anti-hemophilic products despite a satisfactory traditional replacement therapy. For patients with the severe form of hemophilia prophylaxis with intravenous infusion of concentrates is the treatment of choice. Prophylaxis has been shown to significantly reduce arthropathy to limit the frequency of bleeding and to improve the quality of life of patients together with their families or ‘caregivers’.
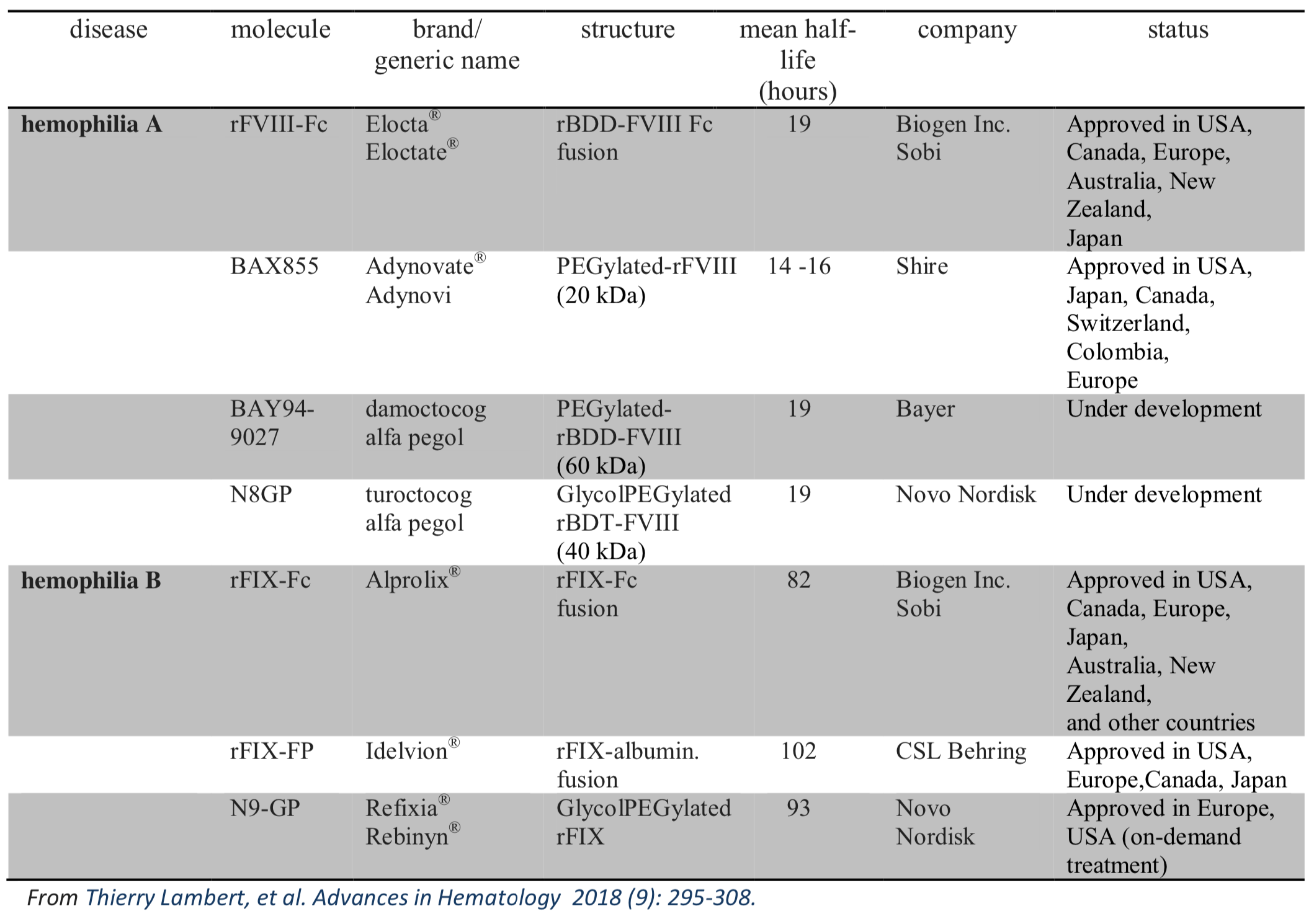
Prophylaxis with traditional recombinant factor VIII (rFVIII) or factor IX (rFIX) concentrates requires regular intravenous infusion of at least two (rFIX) or three (rFVIII) times per week. Nowadays long-acting rFVIII and rFIX products with an extended half-life (EHL) molecules have been approved in Italy and in other Countries while others are still under development. Long-acting rFVIII/rFIX products have a half-life of 1.5-1.8 and 3-5 times respectively if compared to standard concentrates. These new products have the potential to address many of the unmet needs of patients currently treated with traditional replacement therapy. The following table showes the EHL products approved and those under development.
The indications from clinical studies reported on the respective technical sheets of EHL preparations are prophylaxis and treatment of hemophilia. Currently unlike standard regimens there is no consensus about the best dose and the optimal intervals among each infusion in prophylaxis concerning these products.With regard to hemophilia A it is shown that the time spent with a ‘trough level’ of FVIII less than 1% is proportional to the incidence of bleeding while a ‘trough level’ of more than 1% provides greater protection against bleeding mainly against the hemarthroses.
In clinical practice EHL concentrates allow the reduction of the frequency of infusions encourage patients to switch from ‘on demand’ treatment to prophylaxis reduce the use of central venous catheters in children and finally tend to improve their quality of life. In addition it is important to note that EHL products increase the ‘trough level’ at the same dose used for standard preparations prevent spontaneous and sub-clinical bleeding and contribute to maintaining stable the joint functions.The recommended starting dose for adults suffered from hemophilia A who switch to EHL products is based on body weight 2 times or every 3 to 4 days in a week (vs every 2 days with the rFVIII standard prophylaxis).
In children the half-life of the most products is shorter so that also the intervals among the infusions may be shorter. The dose and the time of infusion can be modified according to the bleeding phenotype and to the individual pharmacokinetic (PK) prophile. However in clinical practice PK studies are often avoided especially in children due to numerous blood sampling. This problem could be resolved by the application of PK ‘population’ tests that would take fewer withdrawals with lower costs. PK ‘population’ models allow to study the sources of population variability as well as the variables related to this variability to quantify unexplained components and to process data as a single individual.In adults with haemophilia B the prophylaxis regimen provides the initial dose of once a week versus that of twice a week with standard products. In some well-controlled patients it is possible to extend the dose interval every 10-14 days. Also for children with haemophilia B some products have been shown to have a shorter half-life so that it is reasonable to maintain the initial dose once a week.
It is a matter of debate whether the effective ‘trough level’ with rFIX products should be higher (10- 20 U.I./dl) than the maintenance rate for rFVIII.With regard to PK studies in patients using rFIX concentrates the same considerations as those described for rFVIII products may applied. Also in this case PK ‘population studies’ would seem more applicable in clinical practice.During prophylaxis with EHL products it is useful to perform FVIII/FIX dosage. It is well known that laboratory control can be performed either by a ‘one stage’ coagulation test or by a chromogenic assay. There is evidence that the two methods may result in discrepancies either overestimating or underestimating the blood concentration of both standard and EHL products. The use of one test rather than another is entrusted to the expertise of the specialist and the laboratory crew.
A survey on the switch to EHL products and on perception of inhibitor development shows such perplexity in 57% of patients and in 20% of physicians. Clinical studies do not show any increased risk of inhibitors switching from a plasma derived to a recombinant product or from one rFVIII to another (including full lenght and B-domain-deleted/trunked molecules). Clinical trials on previous treated patients who switched from standard preparations to EHL products have shown the absence or a very low risk of inhibitor development. In clinical practice there is no contraindication for adult hemophiliacs who switch to new products. While awaiting the results of clinical trials in pediatric previous untreated patients it is reasonable to switch to long-acting preparations after the child has exceeded 50 exposures with the standard concentrates. The switch to EHL products is particularly recommended in patients especially adolescents with poor adherence to traditional prophylaxis in those with work/sport activities particularly at risk of trauma and in hemophiliacs with a severe bleeding phenotype.
There is no scientific evidence to counteract the use of EHL preparations in persons with hemophilia who have eradicated the inhibitor by immunotolerance induction provided that a long enough time is spent to avoid recurrence (~ 6-12 months).It should be noted that the finding in some hemophiliacs of a higher level of endogenous von Willebrand factor positively influences the FVIII level of standard and EHL products.
In conclusion the adoption in the clinical practice of EHL anti-haemophilic concentrates is recommended to monitor and adjust the dose of the product and the frequency of the infusions until reaching an effective trough level of maintenance to inform correctly the patient not to disappoint the expectations especially for those who will use rFVIII products. Furthermore in patients with severe bleeding phenotype the prophylaxis regimen with long-acting rFVIIII products may be similar to that used with standard preparations. Finally patients who bleed acutely during prophylaxis with EHL concentrates should use the same product at a reasonable dose in any case lower as compared to that of treatment with traditional products.Although the introduction into the clinical practice of EHL products remains to be evaluated in some aspects (including costs, safety and long-term follow-up of patients) these new preparations currently available and undergoing development represent an extraordinary potential that involves significant changes in the treatment of hemophilia.
Suggested readings
– Thierry Lambert, Gary Benson, Gerry Dolan, et al. Advances in Hematology 2018 (9): 295-308.
– Mancuso ME and Santagostino E. Outcome of Clinical Trials with New Extended Half-Life FVIII/IX Concentrates. J Clin Med 2017; 6: E39.
– Ljung R, Fischer K, Carcao M, et al. Practical considerations in choosing a factor VIII prophylaxis regimen: role of clinical phenotype and trough levels. Thromb Haemost 2016; 115: 913–920.
– Morfini M. The history of clotting factor concentrates pharmacokinetics. J Clin Med 2017; 6: E35.